Introduction:
This page of DIPAC’s website is intended to highlight some of the early scientific studies done in partnership with DIPAC for the sake of continued stewardship of Alaska’s salmon resources, as well as the current salmon sampling program funded by the corporation.
DIPAC has had a dedicated lab space at the Macaulay Salmon Hatchery (MSH) since 1992.
DIPAC experimented with otolith marking pink salmon on a large scale in the late 1980s and early 1990s. DIPAC has been otolith marking 100% of chum salmon and sockeye salmon produced at MSH and Snettisham hatchery since 1996. See details on the beginnings of otolith marking done at DIPAC facilities at this LINK.
For more details on DIPAC’s current release goals & species produced - CLICK HERE.
For more details on DIPAC’s historic releases and returns by species - CLICK HERE.
For more details on DIPAC’s dedication to research, see information on DIPAC’s scholarship page about the Ladd Macaulay Memorial Fellowship program - CLICK HERE.
Otolith Thermal Marks (OTMs):
What is an OTM?
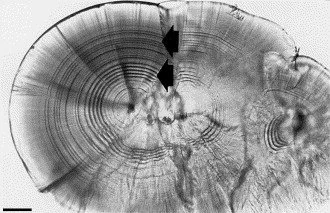
An OTM is a series of visible rings laid on the ear-bone (otolith) of juvenile salmon. DIPAC currently applies OTMs by manipulating the water temperature at both Macaulay and Snettisham Hatcheries. A unique mark is applied to each brood year (i.e. the year in which the eggs were fertilized) allowing us to identify the age and origin of a marked salmon. OTMs are commonly used by hatcheries to better understand different hatchery release strategies and how they impact marine survival.
Please see ADF&G’s website for a detailed look at otolith thermal marking technology at this LINK
Which species and what percentage of DIPAC salmon currently receive OTMs?
Chum - 100% of DIPAC produced chum (since 1996) have an OTM.
Sockeye - 100% of DIPAC produced sockeye (since 1996) have an OTM.
Removing an otolith from a chum salmon that has returned to the hatchery to spawn.
Image of a ThERMALLY MARKED OTOLITH under a microscope - AFTER IT HAS BEEN EXTRACTED FROM A RETURNING SALMON, CLEANED, Mounted to a slide and ground.
Coded Wire Tags (CWTs):
What is a CWT?
A CWT is a small piece of magnetized wire with a unique code etched into it. CWTs are implanted in the snout of juvenile salmon. Being subdermal, they require an additional visible mark to distinguish tagged from untagged fish. This mark comes in the form of removing a fin, specifically the adipose fin, located between the dorsal and caudal (tail) fins. All DIPAC salmon tagged with a CWT have their adipose fin clipped off, but not all clipped fish have a CWT. DIPAC currently uses coded wire tags (CWTs) for its Chinook and coho programs.
Please see ADF&G’s website for a more detailed look at the use of Coded Wire Tags at this LINK
What is DIPAC’s current mark / tag rate?
Coho - 7% of DIPAC coho are tagged with a CWT and 7% are adipose fin clipped (i.e. all clipped DIPAC coho also have a CWT).
Chinook - 20% of DIPAC Chinook are tagged, but 100% are adipose fin clipped (i.e. only one in five clipped DIPAC Chinook will have a CWT). This 100% mark was implemented for the first time on the Chinook that were released from the hatchery in the spring of 2023. The Juneau area will see a significant increase in clipped Chinook returning in 2026.
Check out more on the Northwest Marine Technology Autofish tagging trailer that DIPAC uses here: LINK
In Southeast Alaska, many wild Chinook & coho are also adipose fin clipped and coded wire tagged. Catching a fish with an adipose fin missing, does not necessarily mean you’re catching a hatchery produced fish. Contact ADF&G for more details on these programs.
Hand clipping of An Adipose fin
SPORT CAUGHT CHINOOK SALMON FROM A DIPAC RELEASE SITE - WITH ADIPOSE FIN
SPORT CAUGHT CHINOOK SALMON FROM A DIPAC RELEASE SITE - Without adipose fin
DIPAC Adult Salmon Sampling Programs:
Chum
Since 1995, DIPAC has been sampling and reading chum salmon otoliths at no cost to the State of Alaska. Common property catches of summer chum from D111 (Taku Inlet / Stephen’s Passage) and D115 (Lynn Canal) have been substantially sampled since the mid-1990s and weekly information is provided to ADF&G Commercial Fisheries Managers*. DIPAC currently has two permanent, salaried staff dedicated to research and evaluation, but has had up to 5 staff in the early days while setting up the sampling programs.
DIPAC staff collect sex, weights, scales, and otoliths from the common property fisheries for chum salmon. This data is used in-season to track run status, DIPAC’s contribution to fisheries, and forecasting the next season’s salmon return.
Sockeye
While DIPAC produced sockeye are 100% thermal marked just like the chum, the sampling efforts are quite different between the two. ADF&G has a robust port sampling system in place for southeast Alaska (SEAK) sockeye, meaning DIPAC can calculate common property contributions without needing to take samples ourselves. As such, DIPAC only takes samples from internal cost recovery efforts, while the rest of the run info is derived from ADF&G data.
Chinook
Over the years DIPAC has implemented a variety of tagging strategies in its Chinook programs. These have included differential tag codes for each release site around Juneau and for occasional collaborative experiments. Research and Evaluation staff sample returning adults for tag presence, age, sex, weight, and length right at the Macaulay salmon ladder. Common property Chinook sampling occurs at seafood processors for the commercial catch and through creel surveys for the Sport Fishery. CWT information is provided by ADF&G’s Mark Tag and Age Lab.
Coho
DIPAC’s coho program has utilized a variety of tagging strategies similar to the Chinook program. Most recently, a single tag code has been used for each brood year. Sampling efforts on coho mirror that of Chinook, where internal samples are taken from the Macaulay salmon ladder and common property contributions are provided by ADF&G.
*ADF&G samples the Common Property (CP) catches for Chinook, coho & sockeye. DIPAC performs all of the CP chum salmon otolith sampling in the Juneau area.
More on ADF&G’s Commercial Port Sampling can be found here: LINK
More on ADF&G’s Creel Sampling Programs for Sport Fisheries can be found here: LINK
Miscellaneous Sampling Programs/Lab Work:
Since 1997, the DIPAC Research and Evaluation Lab has been reading all juvenile salmon otoliths for the Southeast Coastal Monitoring Survey (SECM), a joint survey between ADF&G and NOAA.
Since the early 2000’s, DIPAC has been reading all chum salmon otoliths from the Hawk Inlet Test Fishery and the Point Augusta Pink Salmon Index Fishery (in collaboration with NSRAA starting in 2013).
DIPAC’s lab has taken on a variety of contract work throughout the years, mostly in the form of reading scales and otoliths. These collaborations have included other hatchery organizations, state and federal agencies, and educational institutions of all levels.
Research Topics of Interest:
Near Shore Juvenile Interactions:
Like other chum hatchery programs in Alaska, DIPAC produced chum are reared in saltwater net pens for 1-4 months before being released into the wild. While there, the chum are released later and fed to a larger size than their wild counterparts. The additional rearing time allows for better imprinting on the release location. The benefits of this “late-large” program are numerous. Chum reared using this method have higher marine survivals than chum released early and small which are reared in the same conditions. The program also allows wild stocks to out-migrate before DIPAC produced chum, decreasing competition for prey in the nearshore marine environment. DIPAC worked closely with researchers at the Alaska Dept. of Environmental Conservation (ADEC), the University of Alaska Fairbanks, and NOAA’s Auke Bay Laboratories to develop this “late-large” program. LINK to study.
Around the same time as the near shore competition study above, there was another study done in the Icy Strait corridor on chum out-migrating to the Gulf of Alaska. LINK to study. This study looked at bioenergetics of hatchery and wild chum salmon after a few weeks to two months post release as they head out to the greater ocean to grow and feed. From this study, much information was gleaned. One highlight is quoted here: “Interactions between hatchery and wild stocks of juvenile chum salmon do occur temporally in the neritic habitat of Icy Strait, Alaska, particularly during June and July; however, prey resources are large relative to the consumption demands of these stock groups. We estimated that hatchery and wild stocks of juvenile chum salmon consumed only a small fraction of the available prey resource in the habitat. Under the modeling assumptions, this result infers that current levels of hatchery production in southeastern Alaska do not represent a significant impact on the prey resource available in neritic marine habitats.” Since this study occurred, DIPAC has not increased it’s release numbers of chum salmon, and the SECM survey continues to monitor plankton abundance in the Icy Strait corridor.
Ocean Carrying Capacity:
Carrying capacity broadly refers to the amount of biomass an ecosystem is able to sustain. Questions have rightfully been raised about the ability of Alaska’s coastal and offshore ecosystems to sustain wild populations of salmon in addition to hatchery production. Do hatchery produced salmon compete with wild salmon for the same resources at a level that causes harm to wild populations? Or is there enough prey to go around for everyone? The answer is not easily found, and depends on a dizzying number of ecological factors. The Alaska State Legislature’s House Fisheries Committee is a good starting point for those seeking a more objective stance on the current state of affairs: LINK. An overview can be found at the bottom of the page, titled “HFSH ADFG Hatchery Literature Presentation.”
Understanding ocean carrying capacity & salmon’s life at sea is difficult to accomplish. A few research projects & programs of note are listed below for those interested.
North Pacific Anadromous Fish Commission (NPAFC) Salmon Information - LINK - Scroll to bottom for annual hatchery releases by country & species
International Year of the Salmon Expeditions - LINK
Alaska’s Salmon Ocean Ecology Program (SOEP) - LINK
High Ocean Biomass of Salmon and Trends in Alaska Salmon in a Changing Climate. A. Wertheimer & W. Heard, 2018 - LINK
Adult Hatchery Straying:
The topic of hatchery-origin salmon straying into wild systems is an ongoing area of interest in Alaska, particularly regarding pink and chum salmon. A “stray” refers to an adult salmon who spawns in a different stream than the one in which they were born. Straying is a natural part of every salmon species’ reproductive strategy, though stray rates are higher in pink and chum than other species. Alaska’s hatchery program was designed with these life history traits in mind, and policies were developed to minimize risk to wild populations*. The Alaska Hatchery Research Project (AHRP) began sampling chum salmon in SEAK streams all the way back in 2013. While field sampling for the AHRP is now complete, work still needs to be done before results are published. You can follow along with its progress here: LINK
In the meantime, and in the interest of due diligence, DIPAC and other hatchery operators in Alaska are exploring methods of decreasing hatchery stray rates. One method that shows promise is called Integrated Multitrophic Aquaculture (IMTA). In short, IMTA involves growing native kelp beneath our saltwater net pens to absorb waste products from juvenile salmon and act as an attractant to returning adult salmon. Since salmon use smell to find their way back to the stream they were born in, the “smell” of kelp can imprint and serve as a lure for adults that may otherwise decide to stray into a different stream along the way. In addition to these benefits, the kelp being grown underneath the net pens can be harvested with the help of Alaska’s burgeoning kelp farming industry! DIPAC has taken the first steps towards exploring IMTA by assessing water quality at three of its chum sites. If shown suitable for kelp growth, the next steps will be partnering with industry professionals to attempt a pilot project. You can find more information on this topic from our friends at NSRAA: LINK
*ADF&G Policies:
Genetics
Fish Health
Summary of all policies & planning - See the most recent Alaska Fisheries Enhancement Report